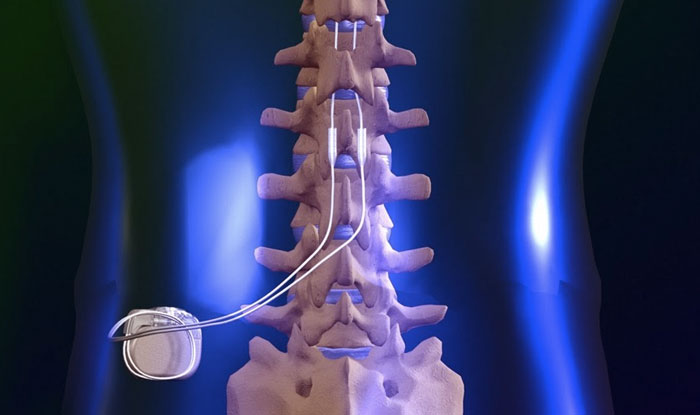
Refer to for product manuals for complete indications, contraindications, warnings, precautions and potential adverse events. Adverse events may result in fluctuations in blood glucose in patients with diabetes. The surgeon opens that capsule to carefully detach the leads without disturbing the spinal cord. This incision exposes the scar capsule that was created when the leads were initially implanted. May include: undesirable change in stimulation (uncomfortable, jolting or shocking) hematoma, epidural hemorrhage, paralysis, seroma, infection, erosion, device malfunction or migration, pain at implant site, loss of pain relief, and other surgical risks. Spinal cord stimulator is a surgically implanted device which relieves pain of the back, ams and legs by masking pain signals before they reach the brain. To remove a spinal cord stimulator that uses paddle leads, surgeons make an incision along the back above a patient’s device. Recharging a rechargeable neurostimulator may result in skin irritation or redness near the implant site. The implanted generator used in spinal cord stimulation has similarities to a cardiac pacemaker. Avoid activities that put stress on the implanted neurostimulation system components. A small device implanted near the spine generates these pulses. Safety and effectiveness has not been established for pediatric use, pregnancy, unborn fetus, or delivery. The first step is a temporary trial to see if it will help control your pain. A preoperative assessment is advised for some patients with diabetes to confirm they are appropriate candidates for surgery. With successful implementation, patients at. Patients with diabetes may have more frequent and severe complications with surgery. Spinal cord stimulator implants provide a non-pharmaceutical alternative by directly targeting the source of pain. An implanted cardiac device (e.g., pacemaker, defibrillator) may damage a neurostimulator, and electrical pulses from the neurostimulator may cause inappropriate response of the cardiac device. Sources of electromagnetic interference (e.g., defibrillation, electrocautery, MRI, RF ablation, and therapeutic ultrasound) can interact with the system, resulting in unexpected changes in stimulation, serious patient injury or death. Spinal cord stimulation (SCS) is indicated as an aid in the management of chronic, intractable pain of the trunk and/or limbs-including unilateral or bilateral pain.ĭiathermy - Energy from diathermy can be transferred through the implanted system and cause tissue damage resulting in severe injury or death.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
